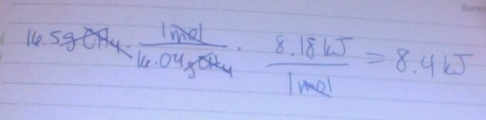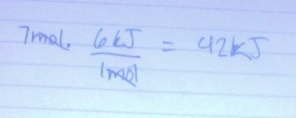Major Points and concepts
Heat of Vaporization:
-41 kJ/mol
-the amount of energy/mol needed to convert between a liquid and a gas
Heat of fusion:
-6 kJ/mol
-the amount of energy/mol needed to convert between solid and liquid
Intramolecular forces: forces within a molecule
Phase Changes: evaporation, boiling, condensation, freezing, melting
Bonds:
-covalent= sharing
-ionic:= between positive and negative
Intermolecular forces: forces between molecules
-Dipole-dipole: partial + and partial - bonds with other partial + and partial -; permanent charge
-Hydrogen bonding: + and - deal with positive H and negative O; permanent charge
-London dispersion: temporary charges of partial + and partial -
Sublimation:
-the process of changing from a solid to a gas without passing through an intermediate liquid phase
Deposition:
-the process of changing from a gas to a solid without passing through an intermediate liquid phase
Alloys:
-substitutional alloy: take one molecule out and put another in
-insistential alloy: put one molecule in without taking another out
ionic solids: stable substances with high melting points that are held together by strong forces existing between oppositely charge ions
molecular solids: tend to melt at relatively low temperature because the intermolecular forces that exist among the molecules are relatively weak
-41 kJ/mol
-the amount of energy/mol needed to convert between a liquid and a gas
Heat of fusion:
-6 kJ/mol
-the amount of energy/mol needed to convert between solid and liquid
Intramolecular forces: forces within a molecule
Phase Changes: evaporation, boiling, condensation, freezing, melting
Bonds:
-covalent= sharing
-ionic:= between positive and negative
Intermolecular forces: forces between molecules
-Dipole-dipole: partial + and partial - bonds with other partial + and partial -; permanent charge
-Hydrogen bonding: + and - deal with positive H and negative O; permanent charge
-London dispersion: temporary charges of partial + and partial -
Sublimation:
-the process of changing from a solid to a gas without passing through an intermediate liquid phase
Deposition:
-the process of changing from a gas to a solid without passing through an intermediate liquid phase
Alloys:
-substitutional alloy: take one molecule out and put another in
-insistential alloy: put one molecule in without taking another out
ionic solids: stable substances with high melting points that are held together by strong forces existing between oppositely charge ions
molecular solids: tend to melt at relatively low temperature because the intermolecular forces that exist among the molecules are relatively weak
Example calculations
1. If you have 16.5g of methane liquid, calculate the energy you will need to convert the liquid to a gas.
2. If you have 5 mol of ice, calculate the energy will you need to convert the ice to water.
Answers:
1.
2. If you have 5 mol of ice, calculate the energy will you need to convert the ice to water.
Answers:
1.
2.