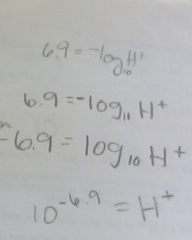major points and concepts
Arrhenius
acid: any compound that increases H+ concentration
base: any compound that increases OH concentration
Bronsted-Lowry
acid: proton-donor
base: proton-acceptor
Lewis
acid: electron pair acceptor
base: electron pair donor
Conjugate acids and bases:
-the conjugate acid of a base is formed when the base gains a proton -the acid and conjugate bases are conjugate pairs and the base and conjugate acids are conjugate pairs
-the reactants are the acids and bases, and the acid corresponds to the conjugate base on the product side of the equation
Strong acids vs. weak acids:
-strong acids will completely ionize
HCl
H2SO4
HNO3
HBr
HI
HClO4
-weak acids will partially ionize
Converting PH and POH
pH +pOH= 14
What is the pOH if the pH was 3.4?
3.4 + pOH=14
pOH=14-3.4
pOH=10.6
acid: any compound that increases H+ concentration
base: any compound that increases OH concentration
Bronsted-Lowry
acid: proton-donor
base: proton-acceptor
Lewis
acid: electron pair acceptor
base: electron pair donor
Conjugate acids and bases:
-the conjugate acid of a base is formed when the base gains a proton -the acid and conjugate bases are conjugate pairs and the base and conjugate acids are conjugate pairs
-the reactants are the acids and bases, and the acid corresponds to the conjugate base on the product side of the equation
Strong acids vs. weak acids:
-strong acids will completely ionize
HCl
H2SO4
HNO3
HBr
HI
HClO4
-weak acids will partially ionize
Converting PH and POH
pH +pOH= 14
What is the pOH if the pH was 3.4?
3.4 + pOH=14
pOH=14-3.4
pOH=10.6