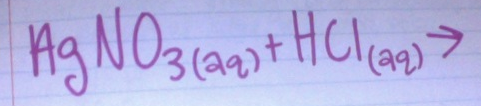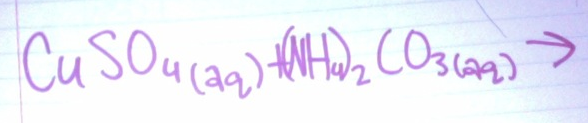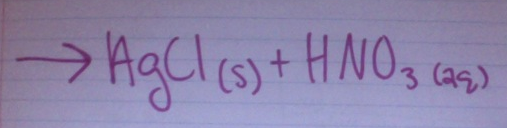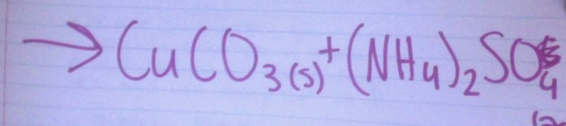Major Points and Concepts
- 4 “driving forces” for reaction
- formation of a solid(ex precipitation)
- formation of water(ex. acid-base or combustion)
- transfer of electron( ex. oxidation- reduction)
- formation of gas(ex. combustion)
- solubility rules
- most nitrate( NO3 -1) are soluble
- most salts of Na+,K+ and NH4+ are soluble
- most chloride salts are soluble-- exception: Ag+,Pb2+ , Hg2 +2
- most sulfates salts are soluble-- exception:Ba2+, Pb2+ , Ca2+
- most hydroxide (OH-)compounds are insoluble-- exception- ions in rule number 2 and Ba/Ca
- most sulfides, carbonate, phosphate are insoluble-- exception- ions in rule number 2
- 3 ways to write chemical equations
- molecular equation
- no charges show
- complete ionic equation
- all the ions that contribute to the formation of an aqueous solution
- net ionic equation
- all the ions that contribute to the formation of a solid
- spectator ion-- the ions that do not contribute to the formation of a solid
- types of reactions:
- combustion
- always has carbon and oxygen gas in the reactant
- always has carbon monoxide or carbon dioxide in the product
- synthesis
- adding two ions to form a compound
- decomposition
- breaking a compound into simpler parts
- single displacement
- one element/compound replaces another one
- double displacement
- elements/compounds switch partners to form a new compound
- acid/base reaction
- there must be both an acid and a bas
- precipitation reaction
- when a solid is formed by two or more aqueous solutions
Example Calculations
Determine whether each or the following compounds are soluble or insoluble:
Write the complete ionic form of this equation:
2 Na3PO4 (aq) + 3 CaCl2 (aq) --> 6 NaCl (aq) + Ca3(PO4)2 (s)
Write the net ionic form of this equation:
Zn (s) + CuSO4 (aq) --> ZnSO4 (aq) + Cu (s)
Determine which type of reaction:
C10H8 + 12 O2 → 10 CO2 + 4 H2O
2 H2O → 2 H2 + O2
Zn + 2 HCl → ZnCl2 + H2
NaCl(aq) + AgNO3(aq) → NaNO3(aq) + AgCl(s)
HA + BOH → H2O + BA
Predict the product:
- AgNO3
- NaCl
- NaCO3
- KPO4
- SOH
Write the complete ionic form of this equation:
2 Na3PO4 (aq) + 3 CaCl2 (aq) --> 6 NaCl (aq) + Ca3(PO4)2 (s)
Write the net ionic form of this equation:
Zn (s) + CuSO4 (aq) --> ZnSO4 (aq) + Cu (s)
Determine which type of reaction:
C10H8 + 12 O2 → 10 CO2 + 4 H2O
2 H2O → 2 H2 + O2
Zn + 2 HCl → ZnCl2 + H2
NaCl(aq) + AgNO3(aq) → NaNO3(aq) + AgCl(s)
HA + BOH → H2O + BA
Predict the product:
Answers:soluble or insoluble:
complete ionic equation:
6 Na+ (aq) + 2 PO43- (aq) + 3 Ca2+ (aq) + 6 Cl- (aq) --> 6 Na+ (aq) + 6 Cl- (aq) + Ca3(PO4)2 (s)
net ionic equation:
Zn (s) + Cu2+ (aq) --> Zn2+ (aq) + Cu (s)
Determine which type of reaction:
Predict the products:
- soluble
- soluble
- soluble
- soluble
- insoluble
complete ionic equation:
6 Na+ (aq) + 2 PO43- (aq) + 3 Ca2+ (aq) + 6 Cl- (aq) --> 6 Na+ (aq) + 6 Cl- (aq) + Ca3(PO4)2 (s)
net ionic equation:
Zn (s) + Cu2+ (aq) --> Zn2+ (aq) + Cu (s)
Determine which type of reaction:
- combustion
- decomposition
- single displacement
- double displacement
- acid base
Predict the products: