Major Points and Concepts
Elements- substances that cannot be decomposed into simpler substances by physical or chemical means
Ions- atoms that either gain or loss one or more electron to form a charged atom
- each element is made up of atoms
- atoms are made up of..
- protons (positive charge)
- neutrons (no charge)
- electrons (negative charge)
- Element names come from many sources...
- languages:
- Latin
- Greek
- German
- places:
- ex: germanium
- ex: francium
- famous scientists:
- ex: einsteinium
- Element symbols:
- the first letter is capitalized
- the second letter is lowercase
- all elements are made of particles called atoms
- all atoms of a given element are identical
- the atoms of a given element are different from those of any other element
- compounds are formed with elements combine atoms
- atoms cannot be created or destroyed, but a chemical reaction can change the way they are grouped together
- compound- a distinct substance that is composed of the atoms of two or more elements and always contain exactly the same relative masses of those elements
- chemical formula- a representation of a molecule in which the symbols for the elements are used to indicate the types of atoms present and subscripts are used to show the relative number of atoms
- rules for writing formulas:
- each atom is represented by its element symbol
- when only one atom of a given type is present, the subscript 1 is not written
- the number of each type of atom is indicated by a subscript written to the right of the symbol
- J.J. Thomson
- showed that atoms of any element can be made to emit tiny negative particles
- the first to consider that there must be a positive charge to balance the negative charges
- Ernest Rutherford
- discovered that by hitting an atom with an alpha particle, sometime there was a deflection, which suggested that there was some sort of core to the atom
- parts of an atom:
- proton(s)
- neutron(s)
- electron(s)
- nuclear atoms- an atom with a dense positive charge around which tiny electrons moved in a space that was otherwise empty
- atomic number- number of protons in the nucleus
- mass number- the sum of the number protons and the number of neutrons
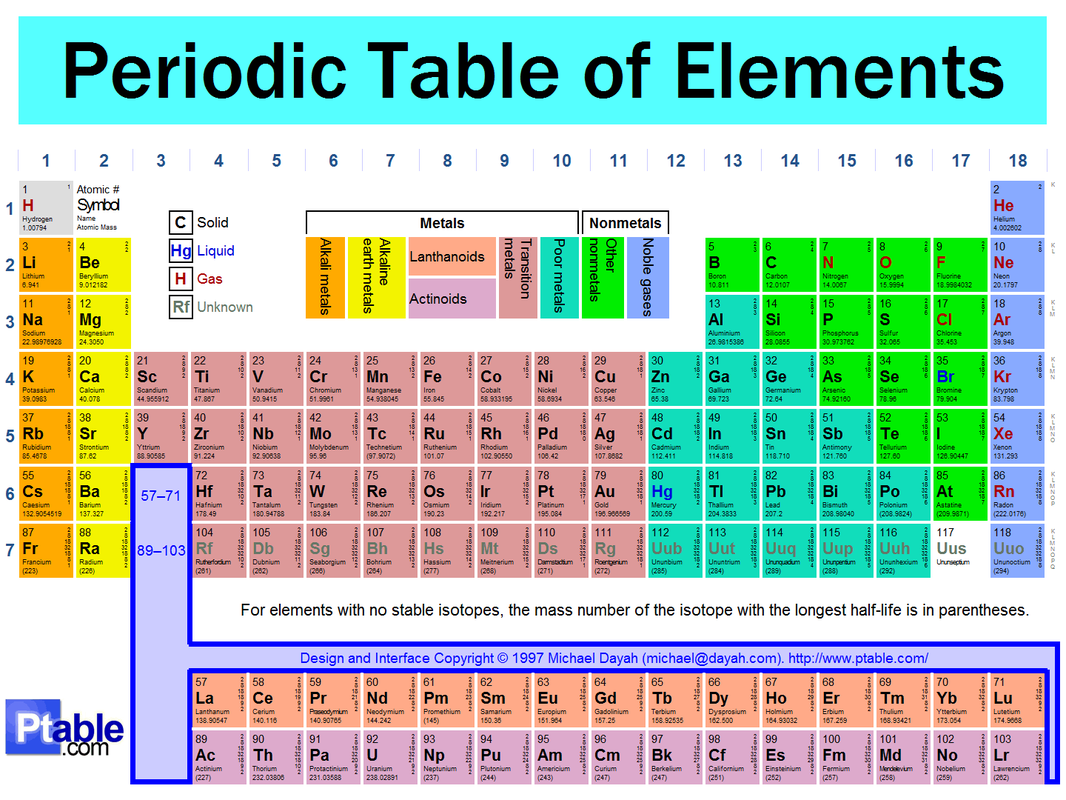
- different sections:
- alkali metals
- alkali earth metals
- translation metals
- halogens
- nobel gases
- groups- vertical columns on the periodic table
- physical properties of metals:
- efficient conduction of heat and electricity
- malleability (they can be hammered into thin sheet)
- ductility (they can be pulled into wires)
- a lustrous (shiny) appearence
- nonmetals- an element that does not exhibit metallic characteristics
- metalloids- an element that has both metallic and nonmetallic properties
Ions- atoms that either gain or loss one or more electron to form a charged atom
- anion- a negatively charged ion
- cation- a positively charged ion
Example Calculations and Problems
Symbols for elements:
- F = Fluorine
- Ne = Neon
- Pt = Platinum
- Au = Gold
- each molecule of a certain compound contains two atoms of nitrogen and five atoms of oxygen---> N2S5
- mass number = number of protons + number of neutrons
- find the number of neutrons for Ca: 40 (mass number) - 20 (protons) = 20 (neutrons)
- the trick for the ionic compound formula is to switch the numbers
- example: Mg-3Na5 would be Mg5Na3.
- what would Mg4 Fe-4 be?---> the 4 and -4 cancel out, to the final compound is MgFe