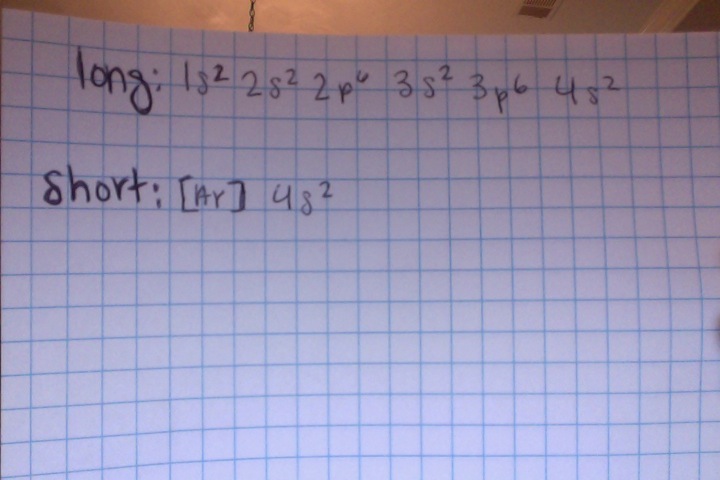Major Points and Concepts
- Rutherford’s Atom
- electrons orbits a small positive core
- the core is made up of protons and neutrons
- most of the atom is empty space
- Electromagnetic radiation
- a form of energy that exhibits wave-like behavior as it travels through space and time
- Different kinds of waves
- radio waves - communication
- microwaves
- infrared - “heat waves”
- visible light - detected by human eyes/ comes from the sun
- speed of light is 2.99*10^(8)m/s
- ultraviolet - causes sunburns
- x-rays - penetrates tissue
- 3 properties of a wave:
- wavelength
- the distance between 2 consecutive peaks or 2 consecutive valleys
- frequency
- indicates how many waves peaks in a given time
- unit: Hz
- speed
- how fast the wave is moving
- Speed=wavelength*frequency
- electromagnetic waves - energy-carrying waves emitted by vibrating electrons
- Wave Mechanical Model of an Atom
- Louis Victor de Broglie and Erwin Schrodinger
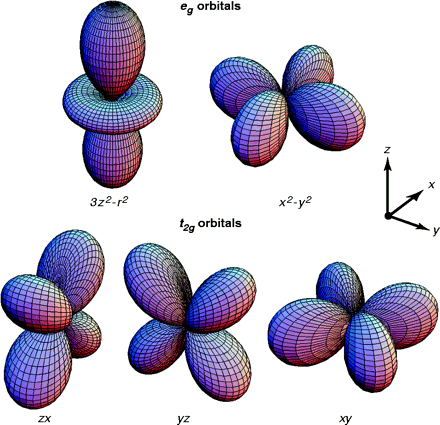
- 4 orbitals:
- S
- P
- D
- F
- suggests the probability of the location of the electron
- states that the more intense the color of a given dot, the more likely the electron can be found
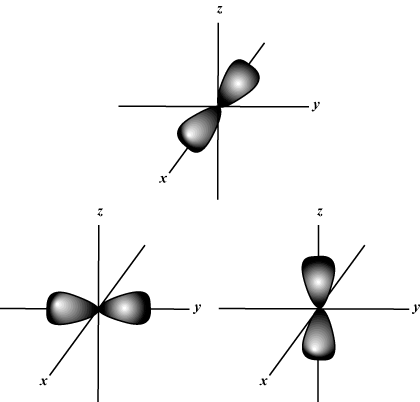
- Orbital- the probability map for the electrons
- labeling
- number tells the principal energy level
- letter tells the shape
- S orbital- sphere
- P orbital- “infinity” shape
- Pauli Exclusion Principle
- one orbital can only have two electrons (but they have to have different spins)
- 3 different kinds of metals on the Periodic Table:
- Alkali metals
- Alkali earth metals
- Transition metal
- Ionization Energy
- the energy required to removed an electron from an individual atom in the gas phase
- most metals have low ionization energy and will give up electron easily
- non-metals have high ionization energy and will not give up electrons easily
Example Calculations and Problems
Identify the orbital:
1st= P orbital
2nd= D orbital
3rd= S orbital
2nd= D orbital
3rd= S orbital
- electron configuration
- do the electron configuration for Ca
- do the electron configuration for NE+2
- construct an orbital diagram for K
- rearrange these elements from smallest to largest based on electronegativity [Li, Rb, K, Na]--> Rb, K, Na, Li
- rearrange these elements from largest to smallest based on ionization [Pt, Ir, Ag, Zn]--> Zn, Ag, Pt, Ir