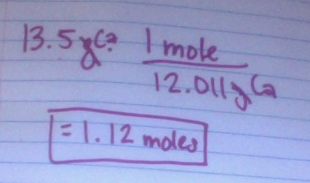Major Points and Concepts
Atomic Mass Unit (amu)
1 amu = 1.66 x 10^-24 g
Mole
- conversion between gram and mole
* molar mass
- conversion between volume and mole
* 22.4 L = 1 mole
- conversion between atom and mole
* avogrado's number
Empirical Formula
- also known as simplified
- use the handy chart
* .7--.9 - round up
* .6 - .4 - multiply by two (multiply all sides)
* .3-- .0- round down
Molecular Formula
molar mass of MF (always given) / molar mass of empirical formula
1 amu = 1.66 x 10^-24 g
Mole
- conversion between gram and mole
* molar mass
- conversion between volume and mole
* 22.4 L = 1 mole
- conversion between atom and mole
* avogrado's number
Empirical Formula
- also known as simplified
- use the handy chart
* .7--.9 - round up
* .6 - .4 - multiply by two (multiply all sides)
* .3-- .0- round down
Molecular Formula
molar mass of MF (always given) / molar mass of empirical formula
Example Calculations
1. Calculate the number of sodium atoms present in a sample that has a madd of 1172.49 amu.
2. Calculate the molar mass of H2O.
3. Find the percentage composition of H2O.
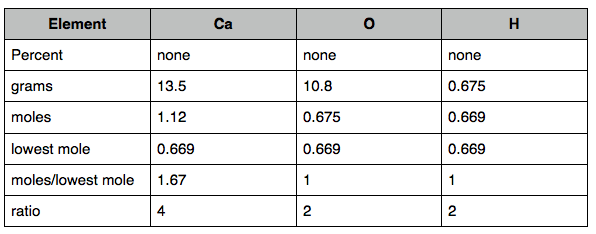
4. A compound was analyzed and found to contain 13.5 g Ca, 10.8 g O, and 0.675 g H. What is the empirical formula of the compound?
5. The empirical formula for vitamin C is C3H4O3. Experimental data indicates that the molecular mass of vitamin C is about 180. What is the molecular formula of vitamin C?
6. Convert 13.5 grams to moles.
ANSWER KEY:
1.
2. Calculate the molar mass of H2O.
3. Find the percentage composition of H2O.
4. A compound was analyzed and found to contain 13.5 g Ca, 10.8 g O, and 0.675 g H. What is the empirical formula of the compound?
5. The empirical formula for vitamin C is C3H4O3. Experimental data indicates that the molecular mass of vitamin C is about 180. What is the molecular formula of vitamin C?
6. Convert 13.5 grams to moles.
ANSWER KEY:
1.
2.
3. H=11.18% O=88.82%
4.
5. C6H8O6
6.
6.